
NIAID supports research on genital herpes and herpes simplex virus (HSV). Studies are underway to develop better treatments for the millions of people suffering from genital herpes. While some scientists are conducting clinical trials to find the best way to use existing drugs, others are studying the biology of HSV. NIAID scientists have identified certain genes and enzymes (proteins) that the virus needs to survive. They are hopeful that drugs aimed at interfering with these viral targets might lead to the design of more effective treatments.
Meanwhile, other researchers are devising methods to control the spread of the virus. Two important means of preventing HSV infection are vaccines and topical microbicides.
Vaccines
Development of a vaccine against genital herpes has been a challenge. Recently, an experimental vaccine designed to prevent genital herpes disease in women, although generally safe and well tolerated, has proved ineffective when tested in a phase III clinical trial called the Women's Herpevac test. NIAID is examing other options of the vaccine and the support of several vaccines in various stages of development
Topical microbicides
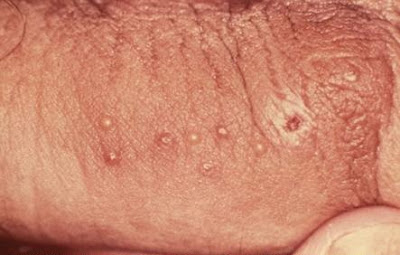
Topical microbicides, preparations containing compounds to kill germs, also are in various stages of development and testing. These include gels, creams or lotions that a woman can insert into the vagina before intercourse to prevent infection. NIAID Sexually Transmitted Infections Clinical Trials Group is conducting a Phase I study to evaluate the safety of a microbicide gel to prevent genital herpes.

A clinical trial support from NIAID shown that once daily suppressive therapy with valacyclovir significantly reduces the risk of transmitting genital herpes to an uninfected partner. This is the first time an antiviral medication reduces the risk of transmission of a sexually transmitted disease. This strategy can help prevent the spread of genital herpes.
No comments:
Post a Comment